THERMODYNAMICS INSULATION
Introduction
The constraints of this project were to design a piece of insulation that was no more than 1 inch thick and contained items from around the house. This material needed to be environmentally friendly and biodegradable While meeting these requirements it needed be as efficient as possible while retaining heat. This material would be put on top of a Styrofoam box that would be heated for 20 minuets with a light bulb. After those 20 minuets were over the light would be turned off and the inner and outside temperatures would be recorded with probes for a period of 20 minuets to find how well the insulation retained heat.
Brainstormin/First Ideas
Our initial idea was to have a layer of tin foil on the outside to deflect heat. Following that would be cardboard. The inside of the cardboard would be filled with cotton balls and/or like materials. After doing some research we found that tin foil was not very effective. We decided to toss that idea and just have cardboard for the outer layer. In addition to cotton balls we decided to fill the insides with the contents of a vacuum cleaner.
Calculation/Documentation
There was a lot of calculating and documenting that went into this project, here is a basic overview of the numbers that we were working with.
The Box:
Inner dimensions: L-15Cm W-15Cm H-17Cm
Volume: .0038 meters cubed
Heat source: 25 Watt light bulb
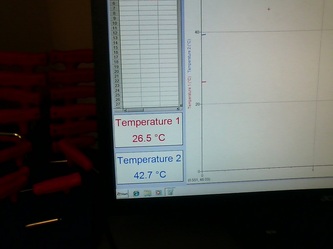
Initial/Maximum inside temperature: 48.1 degrees Celsius
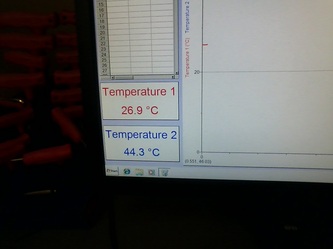
Final/Minimum inside temperature: 36.2 degrees Celsius
Outside/Insulation:
Initial temperature: 22.5 degrees Celsius
Final temperature: 22.5 degrees Celsius
Time:
Heating time: 1200 seconds (20 Minuets)
Cooling time: 1200 seconds (20 Minuets)
Mass of air being heated: .00456kg
Energy gained by air in the box during heating: 113.17J
Energy lost by air in the box during cooling: 54.264J
Net energy retained in box: 59.466J
Tin(max): 48.1J
Tout(max): 22.5J
delta T(max): 25.6J
Comparison Vs. rival teams:
Us: Q (net): 59.466J T (max): 25.600J
Rival 1: Q (net): 17.500J T (max): 9.600J
Rival 2: Q(net): 20.300J T (max): 24.000J
The Box:
Inner dimensions: L-15Cm W-15Cm H-17Cm
Volume: .0038 meters cubed
Heat source: 25 Watt light bulb
Initial/Maximum inside temperature: 48.1 degrees Celsius
Final/Minimum inside temperature: 36.2 degrees Celsius
Outside/Insulation:
Initial temperature: 22.5 degrees Celsius
Final temperature: 22.5 degrees Celsius
Time:
Heating time: 1200 seconds (20 Minuets)
Cooling time: 1200 seconds (20 Minuets)
Mass of air being heated: .00456kg
Energy gained by air in the box during heating: 113.17J
Energy lost by air in the box during cooling: 54.264J
Net energy retained in box: 59.466J
Tin(max): 48.1J
Tout(max): 22.5J
delta T(max): 25.6J
Comparison Vs. rival teams:
Us: Q (net): 59.466J T (max): 25.600J
Rival 1: Q (net): 17.500J T (max): 9.600J
Rival 2: Q(net): 20.300J T (max): 24.000J
Final Design
Our final design consisted of cardboard on the outside. A fairly thick layer of cotton balls followed that. After the cotton balls we used material removed from a central vacuum. Encasing all of this was a large plastic zip-lock whose main function was to compress all of the materials.
Our Total temperature change went from 48.1 to 36.2 degrees Celsius, which is a change of 11.9 degrees Celsius. These results were better than our rivals, but overall they were rather average.
On a more positive note our insulation complied with all of the regulations and was still fairly efficient. It was also very effective at retaining heat, as a little side experiment we let it heat up for longer than 20 minuets, somewhere in the neighborhood of 35 minuets. Our maximum temperature was close to 65 degrees Celsius (nearly 150 degrees Fahrenheit) and still climbing.
Reflection
Looking back on this project I think we did okay for not really knowing any of the properties of the materials we were working with. If I were to do it again there would be some improvements that could be made. For example there should have been a layer of something similar to cotton balls glued to the very outside of the piece of insulation that could form to the heat box and more completely surround it. In doing this project I learned about the phases of design that need to be put in when designing insulation. I also learned about the calculations that must be made when working with thermodynamics.
Images
The following are pictures of our insulating material and our data collections.